Welcome
You need additional expertise or qualified resources for your company or you have a problem related to manufacturing, PR QualityConsultancy can support you with solutions and assessements using Lean SixSigma, GMP-Audits, providing projectmanagement or developing a CMC-development and compliance strategy for new product launches.
Quality and motivation for effective and efficient solutions is the expertise of PR QualityConsultancy. Audits are performed with the goal of gaining value to all parties: Customers get an overview on the real quality standards of their suppliers and the supplier recieves advice and support to improve their product-quality from an independent company.
"No audit-tourism": The overall costs and time-consumption for organizing, planning, travelling and performing qualified audits are too high for all participating parties to be performed without adding value to an organization.
PR QualityConsultancy has the target to contribute value, to be truthful and reliable internal as well as confidential external.
Audits can be performed independent or in combination with your own experts.
Services



About

My company is oriented to improve the quality of my customers in the field of manufacturing and supplier/CMO quality worldwide. This covers process improvement, quality assessment and technical root cause investigation.
About 20 years technical experience in pharmaceutical and chemical technology / engineering, R&D and commercial manufacturing processes as well as quality systems, worldwide supplier-assessment/development and trouble shooting mainly in English or German language.
My educational background is a scientific BSc,MSc / Dipl.-Ing. (Univ.) in BioChemical process engineering from the University of Stuttgart with the main subjects biotechnology, biomedical engineering and chemical reaction engineering. I have 11 years experience as GMP-Lead Auditor worldwide (ECA/APIC-certified; America, Europe, Asia, Australia), hosting authority inspections or for-cause audits by customers, as Lean SixSigma Black Belt and overall 16 years experience in the global pharmaceutical and biotechnology business, with an overview from more than 130 manufacturing sites. This includes ‘TOP 10 – big pharma’ as well as smaller Biotechnology companies
The network can contribute additional expertise and resources to meet customer’s needs. For example, language support for different countries. Our customers are our best proof for quality. We appreciate to be also your contact of choice.
Service
The following service is provided in sterile and non-sterile GMP-manufacturing of bio-/ pharmaceutical API (active pharmaceutical ingredients) and production of FDF (finished dosage forms – liquid/ oral) as well as pharmaceutical excipients and medical devices. Additional this service can be applied in all related supplying businesses (manufacturing equipment and raw materials, clean-rooms, primary and secondary packaging, detergents, disinfectants, disposable equipments etc.)
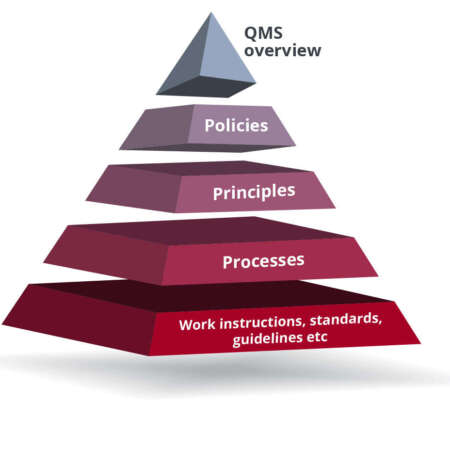
Project Overview
Senior Manager Quality Assurance (Lead): Advisor for Biotech Start-up companies in the field of Cell & Gene Therapy (ATMPs). Technical development strategies for GMP-manufacturing of patient-specific therapies and products with various applications. Manufacturing and release of GMP-batches for clinical trials (phase I,II). Supplier selection and qualification. Development & Improvement of individually adjusted GMP quality system. Authority inspections.
Interim Group-Manager Process Development (DSP): Development of GMP-manufacturing process for RNA-Vaccines (Drug Substance). Data assessement, problem solving, team building with the target of validation readiness (Clinical phase II,III). Technology transfer and scale-up.
Person-in-Plant (PiP): Commercial aseptic manufacturing of a Biosimilar (monoclonal antibody) in prefilled syringes (drug product). Investigation and root-cause assessment of quality issue at CMO for a global organization. Gap-assessment of GMP-compliance and risk assessment. Remediation strategy and monitoring visits at the external manufacturing site.
Senior Consultant: Inspection readiness (US-FDA) compliance of global organization of CAPA-system (EMEA-locations) Strategy, legacy overview, remediation and training of sites.
Lead GMP-Auditor Vaccine / Biopharma CMO-Development – Manufacturing DS/DP including suppliers. (Various customers) USP/DSP Process Development for clinical studies, Technology Transfers and commercial manufacturing. Training/Coaching of Junior-Auditors.
Interim Manager Process Validation / Human vaccine / biotechnological API-manufacturing / mammalian Cell-Culture. CMC-Technology Transfer to CMO – Clinical Phase III – New Product Launch (International organization), different risk assessments, including GMP-audits for supplier qualification and GMP-compliance support. [FDA Guidance for Industry “Process Validation” Stage 2: PPQ-Readiness and Validation]
Compliance-project: Inspection Readiness (FDA, EMA, etc.) – Remediation of Deviation/CAPA-system / subject matter expert / continuous improvement / root-cause investigations / product risk assessment etc. [EN ISO 13485:2012; FDA 21 CFR part 820; Class III sterile medical devices; human implants, etc.]
CMC – Technology Transfer (R&D to GMP-production, Clinical Phase III, New product launch), Risk Assessment, cleaning validation, statistical data assessment in aseptic biopharmaceutical manufacturing FDF – liquid sterile cartriges. [FDA-Guidance for Industry “Process validation” – Stage 1: Quality by Design – ICH Q8/Q9/Q10, etc.]
Senior Consultant: CAPA compliance / legacy review / remediation / subject matter expert in manufacturing of surgery implants, human biomaterials and combination products (class III medical devices), organization change management, investigations continuous improvements etc. (FDA-warning letter / Concent degree) [EN ISO 13485:2012; FDA 21 CFR part 820; Class III sterile medical devices; human implants & biomaterials, etc.]
Implementation of Continuous process verification (CPV) in aseptic manufacturing of biopharmaceutical / biologics drug product (liquid, lyo) according to FDA-Guidance for industry “Process validation” – Stage 3: Process Capability CpK-value, statistical data analysis [ICH Q9/Q10, etc.]
Hosting and guiding different inspections by authorities and for ‘for cause audits` by large and medium pharma customers (CMO business). Including CAPA-Plan assessment.
Post aquisition audits (PAA) at worldwide sites after large company-merger. (Pharmaceuticals, diagnostics, medical devices) – including CAPA-plan assment and Quality Compliance.
Lead GMP-auditor – Compliance and “for-cause” audits of most cell culture media suppliers worldwide (Biopharmaceuticals, ISO 13485) – including CAPA-Plan assessment.
Lead auditor – International supplier qualification & supplier development for divers raw materials and single-use systems (ISO 13485) used in antibody manufacturing (USP/DSP – Biopharmaceuticals – DS/DP) – including CAPA-plan assessment and Quality Compliance.
International GMP-Audits (lead auditor) of contract manufacturers (CMO) for sterile finished dosage forms (cytostatics, biologics, biosimilars, etc. – syringes, vials, ampoules – liquid and lyophilized) including Quality Compliance.
GMP-compliance-Audits (lead auditor) of chemical API manufacturers in China [ICH Q7, etc.].
Senior QA-Manager – Technical QA-supervision of investment projects in pharmaceutical manufacturing (FOYA: factory of the year 2012 – single-use manufacturing facility).
Senior QA-Manager – Technical QA-supervision of cell culture development projects for manufacturing of antibodies/biologics [ICH Q7a, etc.] mammalian CHO manufacturing/ MCB,WCB / suppliers – including all aspects of Quality Compliance.
Manufacturing Scientist – New product launch New product launch in prefilled syringes: Process validation, qualification of equipment, technical trouble shooting, root cause investigations and risk assessments in aseptic biopharmaceutical manufacturing (FDF).
Manufacturing quality scientist – FDA-compliance of different technical projects in commercial pharmaceutical manufacturing, process design and trouble shooting.
Senior manufacturing expert – Continuous process verification (CPV) sterile manufacturing according to FDA-guideline using Minitab- Statistic software
Technical validation expert – Cleaning validation in biopharmaceutical manufacturing of API/Biologics/DS according to ICH Q7a etc.
Lead Auditor – GMP-Audits of manufacturers of disposable systems (polymer systems for biopharmaceutical manufacturing, ISO13485) – including CAPA-plan assessment and Quality Compliance.
QA-Expert –Training of GMP-personnel in risk assessments/ FMEA (aseptic biopharmaceutical processes DS/DP).
Technical qualification expert – Qualification (IQ,OQ,PQ) of pharmaceutical process equipment and utilities (WFI-systems, HVAC, etc.).
Process validation scientist – Product process validations (PPQ) biopharmaceutical manufacturing of Monoclonal antibodies/Mammalian cell culture/API/Biologics/DS (ICH Q7a).
QA-Manager (group lead) – Development of a GMP-system at a smaller biotechnology company – CMO. (Deviations, Change Control, Supplier qualification etc.) including all aspects of quality compliance.
Process Development Scientist – Development of a pervaporative Membrane bioreactor (R&D-Environment). Publication: Journal of membrane science (Elsevier)
Process Development Scientist – Development of a hollow-fiber bioreactor for expansion of hemopoietic stem cells CD34+ / mass transfer simulation. (company scholarship – R&D-Environment)
Process development scientist – Construction of prototypes for car -motor-cooling system (commercial launch).
Process scientist – Thermodynamic heat transfer calculations for pharmaceutical plant engineering (commercial product launch).
Years Experiences
completed projects
My project was a simple & small task, but the persistence and determination of Westy team turned it into an awesome and great project which make me very happy with the result!

In my job as social media strategist, I’ve tried every analytics product on market and when I came across West, I was blown away immediately and great project which make me very!








